- Skype video conferencing camera
- How many people use physiotools
- The binding of isaac rebirth trailer
- Fastrawviewer alternative
- Uiuc stereology microscope
- Cad usd
- Kabhi chup chup rahe
- Leawo itransfer pc world
- Always bon jovi lyrics deutsch
- Apple logic pro x 10-2-1
- Thaniyatha thagam tamil christian song
- Allwinner tablet reset button
- Sleeping dogs cast
- Autodesk viewer free download
- Samsung radianz quartz vs silestone
- Wwe roadblock 2016 matches
- Emerald city confidential steam
- Babydow howrse layouts
- Gfi mailessentials blocking legitimate emails
- #Uiuc stereology microscope skin#
- #Uiuc stereology microscope software#
- #Uiuc stereology microscope plus#
The first scenario will likely offer the most objective solution but is technically more challenging. This may pose a very significant bias that may be circumvented either by performing volumetric analysis or increasing the number or observations to obtain more robust averages per individual. Note that this is valid for counts just as well. Most likely, the same structure, if measured in consecutive sections, will yield different values.
#Uiuc stereology microscope plus#
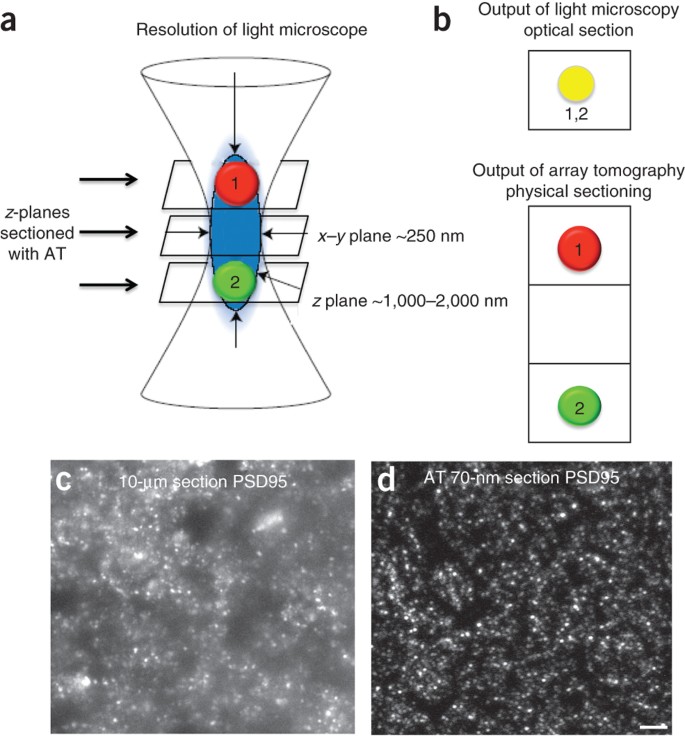
One of the major constraints of performing quantitative analysis on histopathological sections is the fact that the researcher will be essentially determining counts and measurements in an almost two-dimensional object, thus immediately ignoring the potential irregularity of structures that are actually three-dimensional, plus the fact that the sectioning plane may not capture a realistic perspective of its actual size, like the surface of water in relation to an iceberg. Obviously, measuring the size of regular shapes, such as goblet cells and granulocytomas, both tending to be more or less spherical, is far simpler.
#Uiuc stereology microscope skin#
This has been done for instance, to determine changes in goblet cell size in the skin and gills of fish exposed to chemical or pathological challenge by measuring the cells though their longest radial length after histochemical staining with Alcian Blue or PAS (see Yamamoto et al., 2011 Costa et al., 2009b 2013a Costa et al., 2009b Costa et al., 2013a). In fact, besides counts, measuring the size of structures can be a good strategy to obtain quantitative data.
#Uiuc stereology microscope software#
The use of specific histochemical and immunohistochemical stains can yield very precise results to determine the extension of an alteration using image analysis software to detect and quantify light color (as intensity and area of distribution) within the spectrum of a given chromogen or fluorochrome (the latter probably being the best choice for the purpose).

This is particularly useful to quantify traits that are continuous rather than discrete like a melanomacrophage aggregate (e.g., tumors and preneoplastic lesions). Similarly, although perhaps less expeditious without dedicated image analysis software, the researcher can determine the proportion of section area exhibiting an alteration. The values from several sections from the same animal are averaged, thus integrated into a final individual estimate. Similarly, one may count the number of melanomacrophage centers per area of section. As a result, an apoptotic indice (personally, I prefer this term, “indice,” though it is uncanny in the English language, in order to make a distinction from “index”) is obtained, being expressed as the number of apoptotic cells per square millimeter, for example. This area can be estimated through the radius of the optical field, which can be easily estimated in any scale-calibrated microscope (i.e., in which the real distance in microns has been associated to a known distance in pixels for a given magnification). For instance, one may count the number of apoptotic cells per field of observation. The simplest way to obtain a measurement is to perform a count on structures or cells exhibiting some target feature. Even though truly quantitative methods have been widely employed by toxicohistopathologists, especially in fish, mostly on (the few) discrete alterations that can be easily identified and counted. Notwithstanding the variability of protocols, their fundamentals are similar and it is such that will receive focus in the present section. Throughout the available literature there are many approaches to quantify histopathological traits, likely more that can be described here in detail, but the reader is diverted to classic work by Freere and Weibel (1967), where some of the most important technical principles were first compiled and described. In many cases, digital image processing is an important resource in the process.

This implies that a number is produced directly from analysis through counting or measuring the area or volume of a given feature, which is referred to as microscopic stereology. Some histopathological traits can be objectively and directly quantifiable. Costa, in The Handbook of Histopathological Practices in Aquatic Environments, 2018 7.2 Quantitative Histopathology
